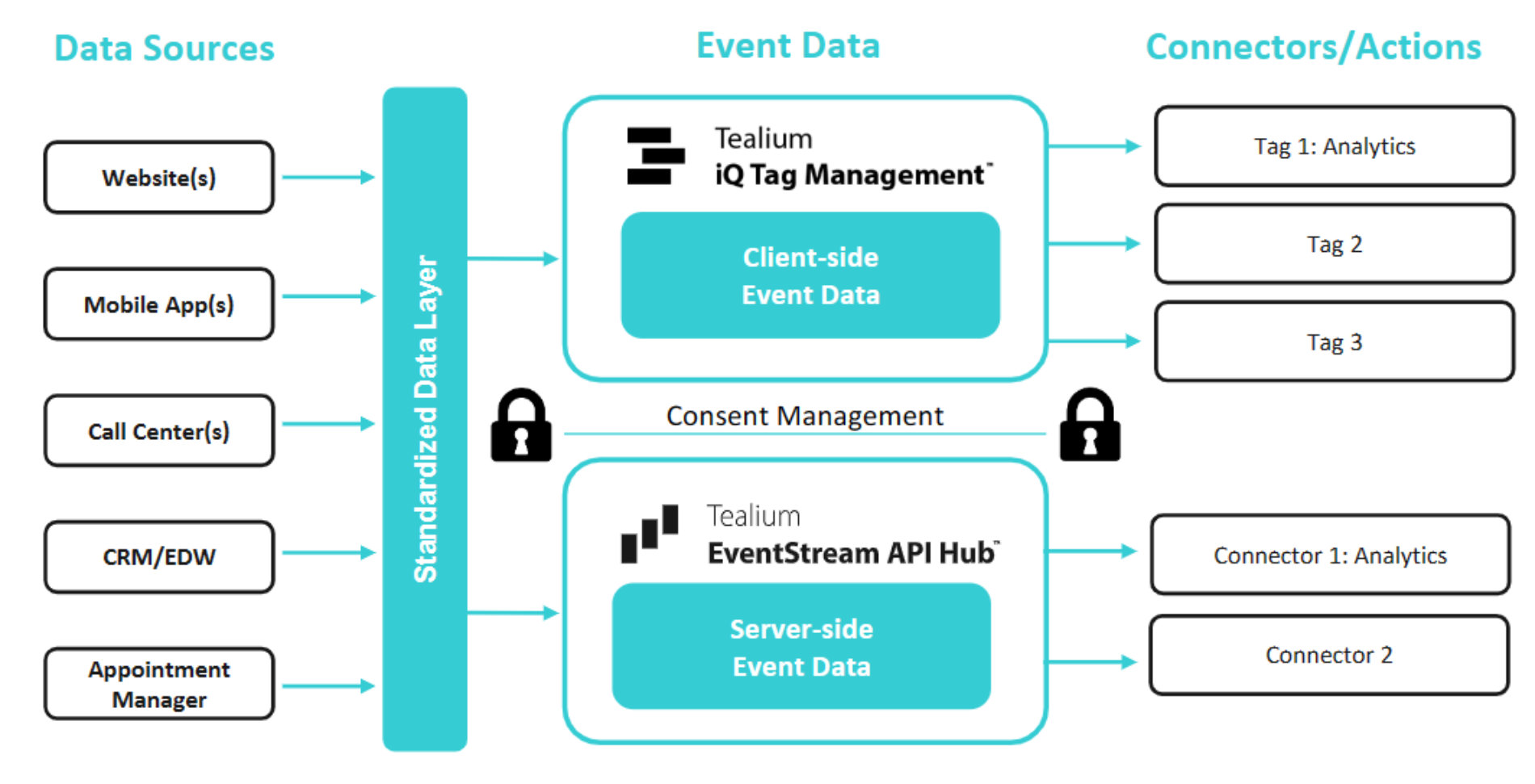
CDPs are connected to robust data collection technologies that are designed to provide secure handling of all PHI that surfaces on the devices patients are using. Encryption and hashing of this data takes place immediately upon these devices, which is critical for HIPAA compliance as it assures the healthcare organization that they become stewards of this data in an appropriate format. Encryption ensures that sensitive information remains secure from the moment it surfaces to the moment that the Healthcare organization takes ownership of it and places it at rest. By implementing encryption protocols within the CDP infrastructure, organizations can comply with HIPAA requirements for safeguarding PHI.
HIPAA requires organizations to implement strict controls to limit access to PHI, and this applies to both internal personnel as well as Business Associates. CDPs can enforce role-based access controls, ensuring that only authorized personnel can access and handle sensitive patient data, or any subset of it that is relevant for the purpose. This reduces the risk of unauthorized data exposure.
CDPs often offer auditing and monitoring capabilities that enable organizations to track data access and changes. These features help maintain an audit trail of who accessed PHI, what changes were made, and when. In addition, CDP profiles will change over time, based on activity or events that take place with the patient journey - these changes can be automated to improve operational efficiency, and the impact of this automation upon the profile is also audited, giving the healthcare organization a complete data lineage and timeline to ensure compliance. By monitoring data activity within the CDP, organizations can identify and respond to any suspicious or unauthorized access, ensuring compliance with HIPAA's security requirements.
HIPAA requires obtaining patient informed consent before collecting and using their PHI. CDPs can facilitate consent management by providing tools that produce digital consent forms, capture the associated privacy preferences chosen, and enforce the implied data flows that they correspond to. This allows organizations to ensure data collection and processing activities align with the specific consents given by patients, enhancing compliance with HIPAA's consent requirements.
HIPAA also mandates proper data retention and disposal practices. CDPs can assist organizations in managing data retention periods and automating data deletion when it is no longer needed. By implementing data lifecycle management within the CDP, organizations can ensure that PHI is retained and disposed of in accordance with HIPAA guidelines.
If an organization uses a CDP provided by a third party vendor, it's crucial to ensure that the vendor itself is HIPAA compliant. Organizations should carefully evaluate the vendor's security measures, data handling practices, and compliance certifications. Working with a HIPAA-compliant CDP vendor helps maintain compliance throughout the data management process because the built-in integrations to Business Associate technology are fully secured and automated to eliminate errors and ensure consistency.